Propane Gas Bottle – How Does Gas in Bottle Go from LPG to Gas – Bottle Gas Propane
In a propane gas bottle (propane bottles), how does the gas in bottle (gas in a cylinder) go from LPG to gas in bottle?
LPG – propane is the gas in bottle (gas in a cylinder) or bottle gas propane.
A propane gas bottle (propane bottles) works when the LPG gas in bottle, stored as a liquid under pressure, turns back from liquid LPG to gas in bottle (gas in a cylinder).
This happens by releasing some of the pressure of the gas in bottles (gas in a cylinder), through use.
Gas in bottles (propane gas bottle) works after being filled with the pressurised LPG (bottle gas propane).
The LPG gas in bottle (gas in a cylinder) is generally stored, as a liquid (bottle gas propane), in steel vessels ranging from a small BBQ propane gas bottle (propane bottles) to larger LPG cylinders and bulk propane tanks.
The LPG gas in bottle (bottle gas propane) turns LPG to gas in bottles as it exits the propane gas bottle (propane bottles), then passing through a gas regulator.
The regulator reduces the LPG pressure of the gas in bottles (gas in a cylinder) to the level used by the appliances, as the pressure in the propane bottles is too high for most applications, at up to 2482 kPa.
Importantly, gas regulators are connected to propane gas bottle (propane bottles) outlet valves because they reduce the propane gas bottle pressure of the gas in bottle (gas in a cylinder) to the much lower 2.75 kPa working pressure.
LPG Gas in Bottle Under Pressure – Propane Gas Bottle – Propane Bottles
 Gas in bottle (gas in a cylinder) for propane bottles or propane tanks is LPG (bottle gas propane) as a liquid under pressure in propane gas bottle (propane bottles), from 0 kPa at -42°C to 1794 kPa at 54°C.
Gas in bottle (gas in a cylinder) for propane bottles or propane tanks is LPG (bottle gas propane) as a liquid under pressure in propane gas bottle (propane bottles), from 0 kPa at -42°C to 1794 kPa at 54°C.
The LPG gas in bottle (bottle gas propane) turns back from liquid LPG (bottle gas propane) to gas in bottle when you release some of the pressure.
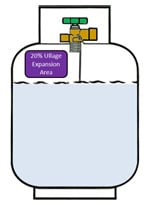
The LPG (bottle gas propane) exists as both liquid and gas in bottle. (See image)
Propane Gas Bottle Works with Liquid & Gas in Bottle
The gas in bottle (gas in a cylinder) is generated when the LPG – propane liquid boils (see image) and turns from LPG to gas in bottle, when you release some of the pressure in the propane gas bottle (bottle gas propane) by using gas (bottle gas propane).

A propane gas bottle (LPG cylinder) works with some of the LPG (bottle gas propane) at the top of the LPG cylinders (propane tanks) as gas in bottle (gas in a cylinder) whilst it is liquid (bottle gas propane) at the bottom. (See image above)
All LPG gases, including propane, are compressed into liquid at relatively low pressures and are also referred to as natural gas liquids – NGL.
Importantly, almost all of the uses for a propane gas bottle involves the use of the gas in bottle (gas in a cylinder), as opposed to the liquefied gas (bottle gas propane).
Propane Gas Bottle – Propane Bottles – LPG Gas in a Cylinder
LPG cylinders may also be called “Propane Tanks”, “Propane Bottles”, “LPG Bottles”, “LPG Gas Bottles” or just “Propane Bottle”.
A propane gas bottle (propane bottles) work by containing both liquid and gas in bottle (gas in a cylinder) because LPG liquefies under relatively low pressure.
A propane gas bottle (propane bottles) is considered low pressure versus high pressure gas cylinders, as used with CNG.
LPG cylinder sizes vary because of application and demand.
A small propane gas bottle is portable, as used in camping.
Show Me My Price
What is a Propane Gas Bottle – Propane Bottles Made of
 An propane gas bottle (propane bottles) is typically made of welded steel, aluminium or composites.
An propane gas bottle (propane bottles) is typically made of welded steel, aluminium or composites.
To start, steel is by far the most common material because it is the easiest to fabricate into a propane gas bottle (propane bottles) and it is a low cost material.
Some propane gas bottle (propane bottles) are made of aluminium or composites since they save on weight.
Aluminium is quite common for a forklift propane gas bottle because of weight relating to safe lifting.
 The use of aluminium keeps the weight down to allow for more gas in bottle (gas in a cylinder) since staying within the safe lifting weight limits is important.
The use of aluminium keeps the weight down to allow for more gas in bottle (gas in a cylinder) since staying within the safe lifting weight limits is important.
The most recent innovation is the composite propane gas bottle (propane bottles).
These propane gas bottle (propane bottles) are typically fiberglass with a high impact plastic outer shell.
Some propane gas bottle (propane bottles) have an inner lining of HDPE whilst other have a thin steel liner because they are impermeable to the gas in bottle (gas in a cylinder).
How LPG (Propane) Liquid Changes from LPG to Gas – Vaporisation
Did you know that every time you turn on one of your gas appliances, the LPG cylinder works (propane gas bottle works) as the liquid LPG (bottle gas propane) starts to boil turning LPG to gas in bottles (gas in a cylinder)?
 Surprisingly, if you could see though the steel of the propane gas bottle, you would also notice that bottle gas propane looks just like water boiling.
Surprisingly, if you could see though the steel of the propane gas bottle, you would also notice that bottle gas propane looks just like water boiling.
The big difference is that it happens at -42°C or -44°F.
Vaporisation is LPG (bottle gas propane) boiling from liquid into its gaseous state as gas in bottle – propane gas bottle (propane bottles).
Vaporisation is how LPG goes from liquid LPG to gas in bottle (gas in a cylinder).
LPG (Propane) Boiling Point
Water boils at 100°C or 212°F, becoming a gas (steam).
In contrast, LPG boils at -42°C or -44°F, becoming gas vapour in the propane gas bottle (propane bottles).
LPG (bottle gas propane) stays liquid because it is under pressure in the propane gas bottle (propane bottles).
As a liquid, bottle gas propane looks a lot like water, if you could see inside the propane gas bottle (propane bottles).
It goes from LPG to gas in bottle (gas in a cylinder) after it boils.
It is colourless and odourless in its natural state.
LPG (Propane) Vaporisation – How Liquid LPG Turns Into Gas in Propane Gas Bottle
Now, a bit more detail on vaporisation in propane gas bottle (propane bottles).
LPG liquid boils and turns LPG to gas in bottle (gas in a cylinder) when you release some of the pressure in the propane gas bottle (propane bottles) by turning on your gas appliance.
 Firstly, to boil, the liquid LPG (bottle gas propane) draws heat from the steel walls of propane gas bottle (propane bottles) which, in turn, works by getting heat from the ambient air. (See image)
Firstly, to boil, the liquid LPG (bottle gas propane) draws heat from the steel walls of propane gas bottle (propane bottles) which, in turn, works by getting heat from the ambient air. (See image)
As with water, the more heat that is applied through the propane gas bottle (propane bottles), the more rapidly bottle gas propane boils, vaporising at a faster rate.
The vapour pressure in the propane gas bottle (propane bottles) also increases with temperature, as explained below.
Because the steel of the propane gas bottle (propane bottles) draw heat from the ambient air heat, cold weather will slow down the rate of vaporisation.
As a result, vaporisation also makes the propane gas bottle (propane bottles) feel colder than the ambient temperature.
Actually using the gas in bottle (gas in a cylinder) makes the propane gas bottle get even colder.
Under the right conditions, frost or ice can even form on the propane gas bottles (propane bottles) because you are using gas (bottle gas propane) very rapidly,
From a practical point of view, it is used just like natural gas, although the LPG (bottle gas propane) energy content is much higher.
As it is a portable gas in bottle (gas in a cylinder), it can be used in vehicles as well as home heating, hot water and cooking.
LPG Pressure and Gas Pressure Regulators
LPG pressure refers to the average force per unit of area that the LPG gas in bottle (gas in a cylinder) exerts on the inside walls of propane gas bottle (propane bottles).
Actual propane gas bottle pressure varies with temperature.
As a result, LPG (bottle gas propane) pressure in the propane gas bottle goes from 0 kPa (0 PSIG or 0 bar) at -43ºC to 2482 kPa (360 PSIG or 24.8 bar) at 70ºC.
 It goes even higher with greater propane gas bottle temperatures.
It goes even higher with greater propane gas bottle temperatures.
The propane gas in bottle (gas in a cylinder) pressure is reduced to at 2.75 kPa, the level used in appliances, by a gas pressure regulator. (see image)
Remember, LPG (bottle gas propane) is a liquefied gas.
So, unlike pressurised gases, the pressure inside the propane bottles will remain the same from full until the vaporistion of the last of the liquid LPG (bottle gas propane).
Then the propane bottles pressure will fall, with the use of the last of the LPG (bottle gas propane) vapour.
Propane bottles pressure measurement is in kilopascals (kPa), pounds per square inch (psi) or “Bar”.
Propane Gas Bottle Pressure Varies with Temperature
As previously mentioned, LPG stored as gas in bottle (gas in a cylinder) is under pressure.
Propane gas bottle (propane bottles) pressure increases with temperature.
(LPG Pressure-Temperature Chart shown)
Propane Bottles – Propane Gas Bottle Pressure Chart
| Temp | Temp | Pressure | Pressure | Pressure |
| ºC | ºF | kPa | PSIG | Bar |
| 70 | 158 | 2482 | 360 | 24.8 |
| 60 | 140 | 2013 | 292 | 20.1 |
| 54 | 130 | 1794 | 257 | 17.9 |
| 43 | 110 | 1358 | 197 | 13.6 |
| 38 | 100 | 1186 | 172 | 11.9 |
| 32 | 90 | 1027 | 149 | 10.3 |
| 27 | 80 | 883 | 128 | 8.8 |
| 16 | 60 | 637 | 92 | 6.4 |
| -1 | 30 | 356 | 51 | 3.6 |
| -18 | 0 | 152 | 24 | 1.5 |
| -29 | -20 | 74 | 11 | 0.7 |
| -43 | -45 | 0 | 0 | 0 |
“Bar” is yet another unit of measure for pressure.
1 Bar = 100 kPa, so it is metric based but not an SI unit of measure.
Propane bottles pressure can vary greatly based on temperature, as shown in the chart.
The level of fill in the propane gas bottle comes into play when the propane gas bottle (propane bottles) is being used, as it affects the rate of vaporisation.
How Much Pressure is in Propane Bottles?
LPG (bottle gas propane) is under pressure, as a liquid, and turns LPG to gas in bottle (gas in a cylinder) when you release some pressure.
LPG cylinders work (propane gas bottle work) with pressure of LPG (bottle gas propane) going from 152 kPa (24 PSIG) at 0°C to 1794 kPa (257 PSIG) at 54°C.
The propane gas bottle (propane bottles) pressure drops to zero at -43ºC (which is just below the boiling point) and the pressure becomes greater at even higher temperatures.
Vaporisation Must Match Consumption When Propane Gas Bottle Work (Propane Bottles Work)
The amount of gas that the appliance or appliances draw from the propane gas bottle (propane bottles) must be matched by the rate of vaporisation when the propane bottles work.
 Switching to larger propane bottles can provide a higher rate of vaporisation of the gas in bottle (gas in a cylinder).
Switching to larger propane bottles can provide a higher rate of vaporisation of the gas in bottle (gas in a cylinder).
Keeping the propane bottles full also increases vaporisation, as there is more liquid gas (bottle gas propane) touching the propane gas bottle (propane bottles) walls.
Heat is absorbed through the propane bottles walls and into the gas in bottle (gas in a cylinder).
The larger the propane gas bottle (propane bottles) or the fuller the propane bottles, the more gas (bottle gas propane) that can be vaporised at a given temperature.
Vaporisation tables are used to match the required vaporisation rates to the corresponding propane bottles size.
The vaporisation tables show the maximum continuous vaporisation rates, in MJ/hr, at different ambient temperatures for each available propane bottles size.
Propane Bottles Work with a Valve
 All propane gas bottle (propane bottles) work with some form of gas valve.
All propane gas bottle (propane bottles) work with some form of gas valve.
A “POL” valve is the standard type of LPG valve in Australia, for propane gas bottle (propane bottles) from 45kg to 210kg.
The POL valve connection is notable for its reverse — left-handed — thread.
A pressure relief valve is incorporated within the POL valve, for safety, in case the pressure of the gas in bottle (gas in a cylinder) gets too high.
Newer LCC27 valves, with added safety features, have been introduced for BBQ size propane gas bottle (propane bottles).
Gas Line Connection – Pigtail
 The gas line pigtail or POL regulator screws into the large female threaded opening on the side of the propane bottles valve.
The gas line pigtail or POL regulator screws into the large female threaded opening on the side of the propane bottles valve.
It is unique in that it has a left-handed or reverse thread. So, to tighten it, you turn the connector anti-clockwise.
Tightening is achieved either with a wrench or by turning a propane gas bottle (propane bottles) hand wheel.
You should always do a soapy water leak test after every reconnection.
Propane Bottles Work by Turning the Gas Valve Hand Wheel
 Propane gas bottle (propane bottles) work by turning the hand wheel anti-clockwise to start the flow of gas in bottle (gas in a cylinder), but you should avoid turning it hard to the open stop.
Propane gas bottle (propane bottles) work by turning the hand wheel anti-clockwise to start the flow of gas in bottle (gas in a cylinder), but you should avoid turning it hard to the open stop.
Located on the top of the valve, the gas valve hand wheel controls the flow of gas, once the connection is secure.
Opening the propane bottles valve 2 or 3 turns is all you need.
You shut the gas off by turning the hand wheel clockwise, tightening firmly by hand only.
CAUTION: Never open the propane bottles valve when unattached
Pressure Relief Valve
 The pressure relief valve is the single most critical safety feature in how propane gas bottle (propane bottles) work.
The pressure relief valve is the single most critical safety feature in how propane gas bottle (propane bottles) work.
It is incorporated within the POL or LCC27 valve and appears as the protrusion opposite the main connection.
The pressure relief valve usually incorporates some kind of plastic dust cover that should be left in place.
Pressure relief valves are designed to relieve excess pressure of the gas in bottle (gas in a cylinder) that might result from overfilling or exposure to excessive heat or fire.
The function of a pressure relief valve is to keep propane bottles from rupturing, in the unlikely event of excessive pressure build-up.
The pressure relief valves are held in the closed position by the force of a powerful spring inside.
As long as the pressure is less than that of the spring, the valve will remain closed.
Propane Bottles (Propane Gas Bottle) Venting Gas
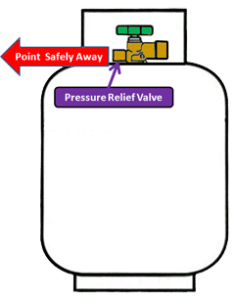 If the pressure rises beyond safe levels, the propane gas bottle (propane bottles) pressure relief valve opens to vent the excess pressure.
If the pressure rises beyond safe levels, the propane gas bottle (propane bottles) pressure relief valve opens to vent the excess pressure.
If this happens, you may hear a hissing sound and see cold gas vapour being released.
Once sufficient propane gas bottle (propane bottles) pressure is released, the valve closes.
If this ever happens, just stay clear of the area and let the gas dissipate.
Having the pressure relief valve releasing gas is a rare event.
Most people will never experience a propane gas bottle (propane bottles) venting.
You should also call your LPG supplier, from a safe location, and advise them that your propane gas bottle (propane bottles) is venting gas.
Do not use your mobile phone, any electrical devices or other ignition sources near a venting propane gas bottle (propane bottles).
Final Thoughts on Propane Bottles Functionality
In conclusion, a propane gas bottle (bottle gas propane) functions unnoticed after evolving over many decades into a very safe storage and gas delivery system.
Propane gas bottle (propane bottles) are also easy to operate and requiring little attention until they need refilling.
- Automated 45kg Gas Bottle Exchange in Ocean Shores - May 11, 2026
- Wet Season Gas Safety: Mullumbimby Home Guide – ELGAS - May 11, 2026
- 45kg LPG Gas Bottles For Your Byron Hinterland Property - May 11, 2026
